Novel Supramolecular CRISPR–Cas9 Carrier Enables More Efficient Genome Editing

The carriers, aminated polyrotaxanes, can flexibly and reversibly bind with Cas9 ribonucleoprotein and protect it from intracellular endosomal degradation
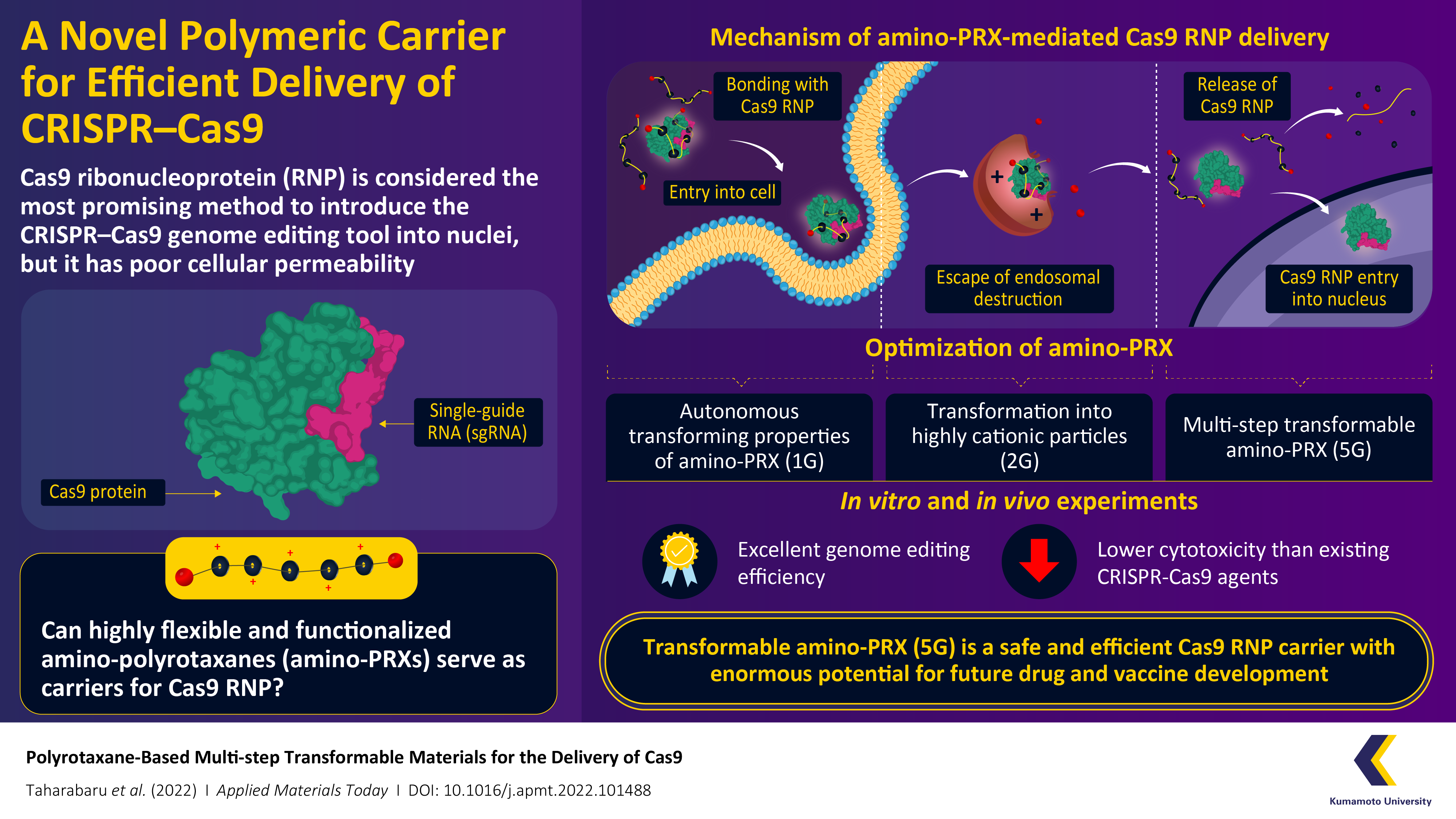
CRISPR-Cas9 is considered a revolutionary gene editing tool, but its applications are limited by a lack of methods by which it can be safely and efficiently delivered into cells. Recently, a research team from Kumamoto University, Japan, have constructed a highly flexible CRISPR-Cas9 carrier using aminated polyrotaxane (PRX) that can not only bind with the unusual structure of Cas9 and carry it into cells, but can also protect it from intracellular degradation by endosomes.Clustered regularly interspaced short palindromic repeats (CRISPR) and their accompanying protein, CRISPR-associated protein 9 (Cas9), made international headlines a few years ago as a game-changing genome editing system. Consisting of Cas9 and strand of genetic material known as a single-guide RNA (sgRNA), the system can target specific regions of DNA and function as ‘molecular scissors’ to make precise edits. The direct delivery of Cas9–sgRNA complexes, i.e. Cas9 ribonucleoproteins (RNPs) into the nucleus of the cell is considered the safest and most efficient way to achieve genome editing. However, the Cas9 RNP has poor cellular permeability, and thus requires a carrier molecule to transport it past the first hurdle of the cell membrane before it can get to the cell nucleus. These carriers need to bind with Cas9 RNP, carry it into the cell, prevent its degradation by intracellular organelles called ‘endosomes,’ and finally release it without causing any changes to its structure.
In a recent paper published in June 2022 in Volume 27 of Applied Materials Today, a research team from Kumamoto University has developed a transformable polyrotaxane (PRX) carrier that can facilitate genome editing using Cas9RNP with high efficiency and usability. “While there have been some PRX-based drug carriers for nucleic acids and proteins reported before, this is the first report on PRX-based Cas9 RNP carrier. Moreover, our findings describe how to precisely control intracellular dynamics across multiple steps. This will prove invaluable for future research in this direction,” says Professor Keiichi Motoyama, a corresponding author of the paper.
For their novel carrier, the research team focused on PRX with amine groups, i.e. amino-PRX, and went through multiple rounds of development and optimization before achieving their final product. For example, the first generation (1G) of their carrier molecules, exploited the autonomous transforming properties of amino-PRX to efficiently complex it with Cas9 RNP and enable its delivery past the cell membrane. The second generation (2G) worked towards endosome-escape. This was achieved via the transformation of the amino-groups in amino-PRX into highly cationic (positively charged) particles within the endosome, which resulted in the rupturing of the endosome and the escape of Cas9 RNP–amino-PRX. The next few generations addressed problems relating to the release of Cas9 once the complex had escaped the endosome. Finally, they developed the fifth generation (5G) multi-step transformable amino-PRX carrier that could precisely and efficiently deliver Cas9 RNP into the cell nucleus. The research team further performed in vitro and in vivo experiments to confirm the cytotoxicity of the system, as well as its genome editing efficiency. “Our delivery system has a low cytotoxicity and its genome editing activity is equal to the current most efficient system on the market,” reveals Associate Professor Taishi Higashi of Kumamoto University, who is the other corresponding author of the study. “Moreover, our multiple attempts at optimizing the delivery system across generations offers important information on the types and positions of various biodegradable groups and amino groups that can be used in such a system to further customize and adapt their properties.”
The autonomous action, multi-step transformable properties, and low cytotoxicity of the 5G amino-PRX carrier make it an enormously promising candidate for the safe and efficient delivery of Cas9 RNP. These findings could furthermore be applied for the delivery of a wide range of molecules, such as enzymes, antibodies, and small interfering RNA (siRNA), thereby making this novel carrier a significant achievement in the field of drug and vaccine development.
About Professor Keiichi Motoyama and Associate Professor Taishi Higashi
Dr. Keiichi Motoyama is a Professor at the Department of Physical Pharmaceutics, Kumamoto University, Japan. His research interests include cyclodextrin drug carriers, drug delivery systems, supramolecular host–guest interaction, and sustained drug release. He has 174 publications to his credit and has been cited 2453 times.
Dr. Taishi Higashi is an Associate Professor of Priority Organization for Innovation and Excellence at Kumamoto University, Japan. His research is focused on cyclodextrin-based supramolecular pharmaceutical sciences. Dr. Higashi has published over 100 papers and has won eight prestigious awards, including the Pharmaceutical Society of Japan Award for Young Scientists in 2018.
[Funders]
- The Leading Initiative for Excellent Young Researchers (LEADER), MEXT
- The Naito Foundation
- Adaptable and Seamless Technology Transfer Program through Target-Driven R&D (A-STEP), JST (20338492)
- Grant-in-Aid for JSPS Research Fellow, JSPS (21J20999)