Novel Vaccine May Yield Affordable Solution to Lipid Disorders

Scientists develop a new candidate vaccine that improves obesity-related disorders and may be economical
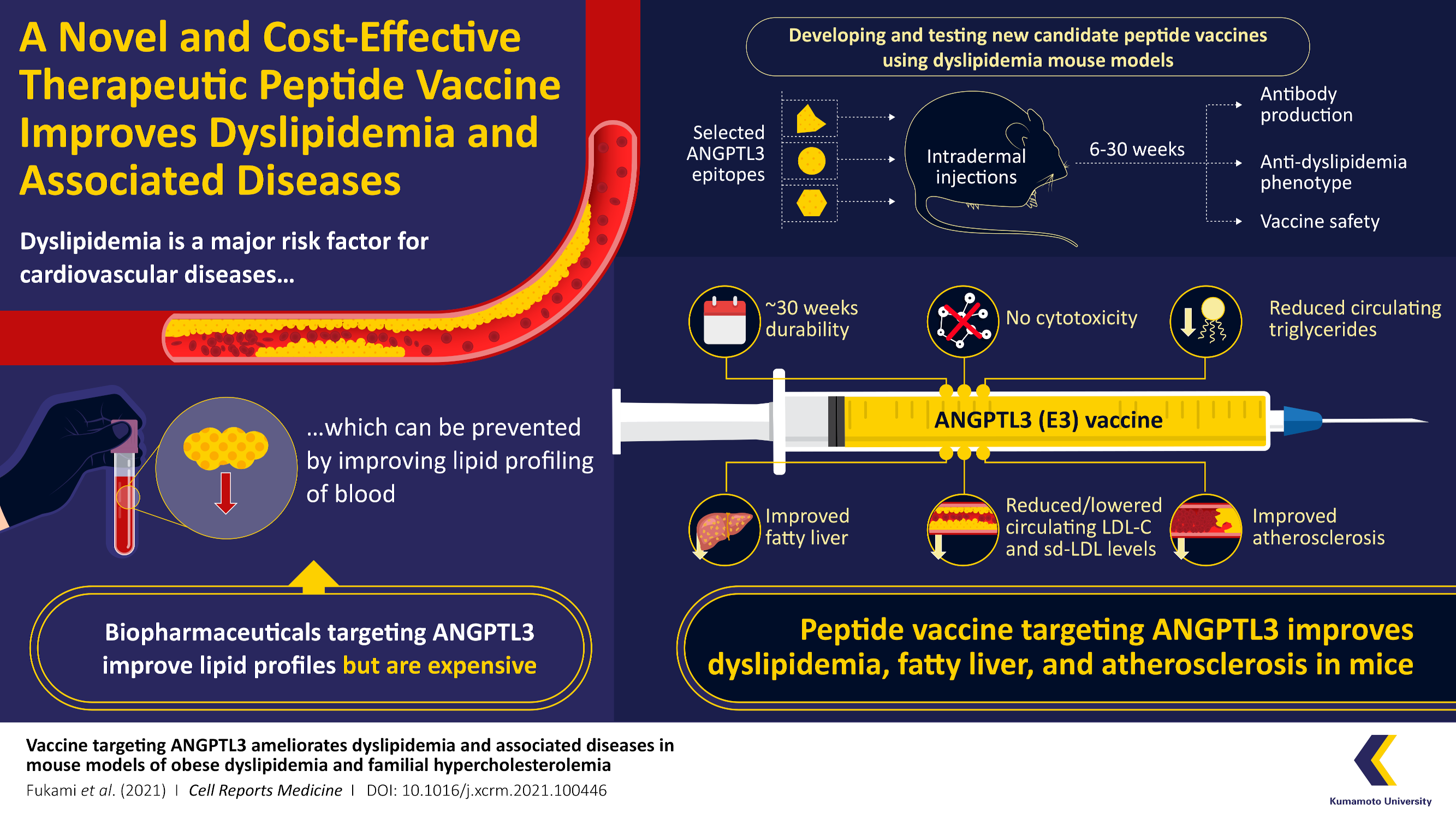
Dyslipidemia is a major risk factor for cardiovascular disease, which can significantly shorten life expectancy. Recently, Prof. Yuichi Oike and his team of scientists at Kumamoto University, Japan, have successfully developed a peptide vaccine that can alleviate conditions of dyslipidemia and proven its efficacy in mice. The new vaccine could become one of the most cost-effective treatments for obesity/cholesterol-related disorders like atherosclerosis and fatty liver disease.Cardiovascular disease (CVD) is implicated in nearly 32% of all fatalities worldwide. Increased levels of low-density lipoprotein-cholesterol (LDL-C) and triglycerides (TG) characterize dyslipidemia, a major risk factor for CVD. These can hasten the progression of fatty liver disease, which can lead to liver cancer in the long run. With the global frequency of fatty liver disease on the rise, the market should soon be flooded with expensive drugs known as "biopharmaceuticals" that are currently under clinical trial. These drugs, however, will require long-term treatment which can be a big obstacle to global health-care coverage. Therefore, scientists are currently focusing their efforts on developing new therapeutic agents that are both effective and cost-efficient.
In their new study, which was published in the Cell Reports Medicine, Professor Yuichi Oike and his team developed a novel peptide vaccine to tackle obesity-associated dyslipidemia and investigated its effects on obese mice models. The paper was published in Volume 2 Issue 11 of the journal on 16 November 2021.
To develop the vaccine, they chose to target angiopoietin-like protein 3 (ANGPTL3), which has recently attracted attention in strategies aimed at decreasing LDL-C and TG levels in the blood. The results of the ANGPTL3 peptide vaccine were validated in their trials on mice, which confirmed efficacy for roughly 6 months with no reported toxicity, yielding evidence that the novel treatment is safe and without side effects.
Peptide vaccines are well-known for being cheaper to manufacture than biopharmaceuticals, making them preferable in terms of medical economy. To meet this need, the team developed vaccines targeting three peptides (or short chains of amino acids) from ANGPTL3, namely, epitope 1 (E1), E2, and E3. The peptide vaccines were then injected into a mouse model of obesity-induced dyslipidemia. The researchers were thrilled to see that the E3 peptide vaccine effectively reduced dyslipidemia in the mice and produced antibodies that lasted 6 months. “The peptide vaccine selectively induces antibody-mediated immunity to produce neutralizing antibodies suppressing ANGPTL3 function”, says Prof Oike.
Because LDL-Cs easily get deposited in the walls of blood vessels and obstruct blood flow, they are commonly referred to as "bad cholesterol". This obstruction can cause atherosclerosis and result in heart failure. To see if their vaccine worked against this condition, the researchers administered E3 into familial severe hypercholesterolemia model mice with a high-cholesterol diet and discovered that it improved dyslipidemia and atherosclerosis.
When asked how this vaccine works and whether similar benefits will also be noticed in humans, Prof. Oike states, "E3 vaccination in mice decreases circulating levels of LDL-C and TG in the blood. Since the mouse E3 sequence is identical to the corresponding human sequence, E3 could potentially be investigated in clinical studies”.
Prof. Oike and his colleagues believe that this vaccination will help patients in economically disadvantaged areas who require long-term therapy for dyslipidemia or related conditions. "Expensive antibody treatments for dyslipidemia require monthly injections, whereas our vaccination is effective for up to 6 months", he explains. "The vaccine is projected to become one of the new economical therapy choices for dyslipidemia and dyslipidemia-related disorders", he adds.
The authors plan to conduct preclinical research to test the vaccine's efficacy and safety, as well as make improvements to prolong its effectiveness. Prof. Oike continues, "We will modify the vaccine formulation and optimize the dosage and administration for clinical trials. Re-immunization every 5 or 6 months could maintain the therapeutic efficacy of the E3 vaccine therapy, and more investigation is necessary to see if a vaccination booster would extend its durability."
About Professor Yuichi Oike
Professor Yuichi Oike currently works at the Kumamoto University's Graduate School of Life Sciences. He graduated from Kumamoto University with a Ph.D. in Medicine in 1999. His main research focuses on identifying the molecular mechanisms of aging and related diseases including obesity, diabetes, dyslipidemia, atherosclerotic diseases, heart failure, and cancer. He is a medical doctor and his major is cardiology and metabolism. He is also the Director of the Japanese Society of Anti-aging Medicine and the Japan Anti-aging Foundation. He is one of the few researchers who have identified ANGPTL family genes and has published many papers regarding the roles of ANGPTLs in physiology and pathophysiology. He has received numerous research awards and grants and is actively involved in joint research worldwide.[Source]
Fukami, H., Morinaga, J., Nakagami, H., Hayashi, H., Okadome, Y., Matsunaga, E., … Oike, Y. (2021). Vaccine targeting ANGPTL3 ameliorates dyslipidemia and associated diseases in mouse models of obese dyslipidemia and familial hypercholesterolemia. Cell Reports Medicine, 2(11), 100446. doi:10.1016/j.xcrm.2021.100446
[Funders]
- JSPS KAKENHI (21K20935)
- Center for Metabolic Regulation of Healthy Aging of Kumamoto University
[Usage Restrictions]
The authors request that a reference to this release be included when using the media contained within this release elsewhere.