How blood and lymph vessels remain separated after development
Researchers from Kumamoto University (Japan) have clarified the mechanism by which blood and lymph vessels remain segregated from one another after development. The characteristics and structures of these two vessel types are very similar, and how they maintain separation has remained unexplained for many years. In this study, researchers found that the molecule Folliculin (FLCN) in vascular endothelial cells acts as a gatekeeper to maintain that separation.
Blood and lymphatic vessels form independent networks until the final confluence at the left and right venous angles in the neck. Blood vessels act as a pipeline that transports oxygen from the lungs to tissues throughout the body. Lymphatic vessels, on the other hand, take in tissue fluids that cannot be collected by blood vessels and act as part of the immune system. However, blood and lymph vessel characteristics and structures are very similar so how they maintain separation from each other has long been a mystery. The elucidation of this mechanism could provide the foundation for the development of novel therapeutic approaches to human lymphatic disorders. For example, the creation of a bypass (a venous-lymphatic shunt) allowing clogged lymphatic flow to return directly to veins would become an innovative therapy for postoperative lymphedema. Thus, researchers around the world have been looking into blood-lymphatic vessel separation mechanisms, but it is still not completely understood.
In this study, researchers first focused on FLCN, the causative gene of a genetic disorder with typical symptoms of multiple lung cysts, kidney cancer, and benign skin tumors (Birt-Hogg-Dube syndrome). They found that mice that were missing the Flcn gene in vascular endothelial cells had abnormal anastomosis between blood and lymph vessels that resulted in fetal death. Furthermore, even in postnatal mice with normal blood and lymphatic vessel separation, induced Flcn gene depletion in endothelial cells caused abnormal anastomosis between the two vessel types.
Flcn deficiency gives rise to lymphatic-biased vascular endothelial cells. These cells cause anastomosis between blood and lymphatic vessels. Prox1* is a central transcription factor in the regulation of lymphangiogenesis and its expression is suppressed by Flcn in venous endothelial cells to maintain the identity of vascular endothelial cells. However, when Flcn regulation in vascular endothelial cells is lost, Prox1 becomes expressed, making venous endothelial cells lymphatic biased. Researchers also found that deletion of Flcn results in the translocation of the transcription factor Tfe3, which is normally in the cytoplasm, into the nucleus where it directly upregulated Prox1 expression. In fact, when Flcn knockout mice were crossed with Tfe3 knockout mice, Prox1 was not expressed in the absence of both Tfe3 and Flcn, and lymphatic biased venous endothelial cells were not seen. These results demonstrate that FLCN acts as a gatekeeper that regulates the plasticity and maintains the separation of blood and lymphatic vessels.
"These results are of great academic importance in that they have clarified a biological question that has gone unanswered for many years: Why do two very similar circulatory systems in the body, blood and lymph vessels, form and maintain independent networks? From a clinical point of view, this has the potential to provide clues on the mechanism of cancer metastasis and can potentially be developed into a treatment for lymphedema," said Associate Professor Masaya Baba, who led the study. "In cancer surgery, lymph node removal can result in serious edema in the upper and lower limbs due to decreased ability to drain lymphatic fluid. Physical therapy, elastic stockings, and lymphovenous anastomosis are listed as treatment methods, but even with skilled microsurgery techniques, it is difficult to treat sufficiently. Out of this research, we envision a future innovative treatment for lymphedema where we can pharmaceutically intervene in the signaling pathway of FLCN to artificially create a shunt that locally connects veins and lymph vessels."
This research was posted online in Nature Communications on 9 December 2020 and was selected as one of Nature Communications Editor’s Highlights. The link to the featured article can be found in the translational and clinical research page.
*Prox1 (prospero-related homeobox 1) is a transcription factor that is essential for the development of lymphatic endothelial cells from vascular endothelium cells during development. It is normally expressed specifically in lymphatic endothelial cells and Prox1 is a master gene for the formation of lymphatic vessels. Artificially expressing Prox1 in blood vessels is known to induce their differentiation into lymphatic vessels.
Source:
I. Tai-Nagara, Y. Hasumi, D. Kusumoto, H. Hasumi, K. Okabe, T. Ando, F. Matsuzaki, F. Itoh, H. Saya, C. Liu, W. Li, Y. Mukouyama, W. Marston Linehan, X. Liu, M. Hirashima, Y. Suzuki, S. Funasaki, Y. Satou, M. Furuya, M. Baba, and Y. Kubota, “Blood and lymphatic systems are segregated by the FLCN tumor suppressor,” Nature Communications, vol. 11, no. 1, Dec. 2020.
[Publication URL]
https://www.nature.com/articles/s41467-020-20156-6
DOI: 10.1038/s41467-020-20156-6
[Fund(s)]
1) Grants-in-Aid for Specially Promoted Research from the Ministry of Education Culture Sports Science and Technology of Japan, 2) Japan Agency for Medical Research and Development PRIME, 3) Inamori Foundation, 4) The Kao Foundation for Arts and Culture, 5) Takeda Science Foundation, 6) Mochida Memorial Foundation, 7) The Mitsubishi Foundation, 8) The Cell Science Research Foundation, 9) SENSHIN Medical Research Foundation, 10) The Sumitomo Foundation, 11) Daiichi Sankyo Foundation of Life Science, 12) The Naito Foundation, 13) The Uehara Memorial Foundation, 14) The Joint Usage/Research Center Program of the Advanced Medical Research Center, Yokohama City University, 15) Toray Science Foundation, 16) Intramural Research Program of the NIH National Cancer Institute Center for Cancer Research
[Numbers]
1) 22122002, 25713059, 15K15089, 18H05042, 18K19553, 17K15625, 18K16997, 18H02938, 18K19619, 19K07389, 2) JP19gm6210017h0001, JP20gm6210017h0002, 3-16) NA
[Media 1]
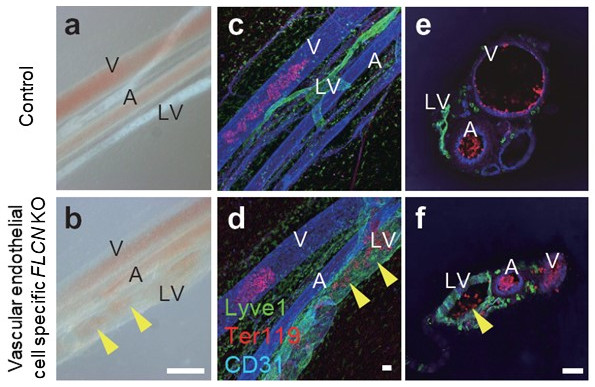
Abnormal [Blood-Lymphatic Vessel] Anastomosis in Vascular Endothelial Specific Flcn Deficient Mice
Mesenteric images in wild-type (a, c, e) and vascular endothelium-specific Flcn-deficient (b, d, f) mice. In endothelium-specific Flcn-deficient mice, blood vessels and lymphatic vessels are abnormally anastomosed, and red blood cells flow into the lymphatic vessels.
CREDIT:
Associate Professor Masaya Baba
USAGE RESTRICTIONS:
Adapted from Fig. 1(s,t,u,v,w,x) of Tai-Nagara, I. et.al., Nature Communications 2020. These images have been modified for use in this press release.
This image is made available under the terms of CC by 4.0. Any further distribution of this work must follow the terms of this license.
[Media 2]
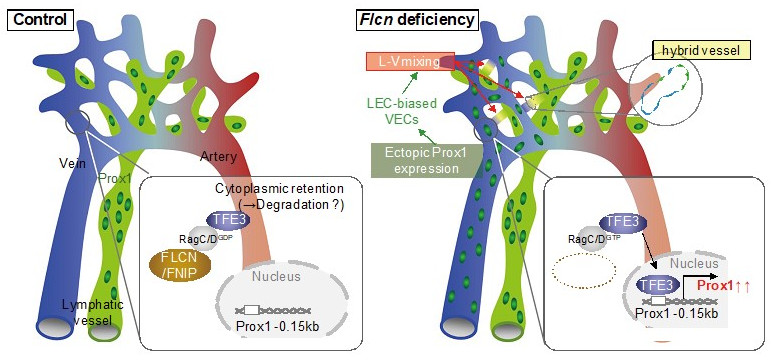
Mechanisms that maintain the separation of blood vessels and lymph vessels
Normally, FLCN keeps the transcription factor TFE3 in the cytoplasm and suppresses the expression of Prox1 in veins. When FLCN is deleted, TFE3 migrates into the nucleus and Prox1 is expressed resulting in the appearance of venous endothelial cells that resemble lymphatic vessels. In other words, FLCN acts as a gatekeeper that regulates the plasticity of blood and lymphatic vessels, maintaining the separation between them.
CREDIT:
Associate Professor Masaya Baba
USAGE RESTRICTIONS:
Adapted from Fig. 7(o) of Tai-Nagara, I. et.al., Nature Communications 2020. These images have been modified for use in this press release.
This image is made available under the terms of CC by 4.0. Any further distribution of this work must follow the terms of this license.
