Bacterial infection stresses hematopoietic stem cells Infection-induced hematopoietic stem cell stress may lead to anemia or leukemia.
It has been thought that only immune cells would act as the line of defense during bacterial infection. However, recent research has revealed that hematopoietic stem cells, cells that create all other blood cells throughout an individual’s lifetime, are also able to respond to the infection. A collaboration between researchers from Japan and Switzerland found that bacterial infection activates hematopoietic stem cells in the bone marrow and significantly reduces their ability to produce blood by forcibly inducing proliferation. These findings indicate that bacterial infections might trigger dysregulation of blood formation, such as that found in anemia or leukemia. This information is important to consider in the development of prevention methods for blood diseases.Background: Bacterial Infection and the Associated Immune Reaction
When a person becomes infected with a virus or bacteria, immune cells in the blood or lymph react to the infection. Some of these immune cells use “sensors” on their surfaces, called Toll-like receptors (TLR), to distinguish invading pathogens from molecules that are expressed by the host. By doing so, they can attack and ultimately destroy the pathogen thereby protecting the body without attacking their own cells.
Bone marrow contains hematopoietic stem cells which create blood cells such as lymphocytes and erythrocytes throughout life. When infection occurs, a large number of immune cells are activated and consumed. It therefore becomes necessary to replenish these immune cells by increasing blood production in bone marrow. Recent studies have revealed that immune cells are not the only cells that detect the danger signals associated with infection. Hematopoietic stem cells also identify these signals and use them to adjust blood production. However, little was known about how hematopoietic stem cells respond to bacterial infection and how it affected their function.
Proof: Hematopoietic Stem Cell Response to Bacterial Infection
The researchers from Kumamoto University and the University of Zurich analyzed the role of TLRs in hematopoietic stem cells upon bacterial infection, given that both immune cells and hematopoietic stem cells have TLRs. Lipopolysaccharide (LPS), one of the key molecules found in the outer membrane of gram negative bacteria, is known to cause sepsis. LPS was given to laboratory animals to generate a bacterial infection model. They also analyzed the detailed role of TLRs in hematopoietic stem cell regulation by combining genetically modified animals that do not have TLR and related molecules, or agents that inhibit these molecules.
As a result, the researchers found that LPSs spread throughout the body, with some eventually reaching the bone marrow. This stimulated the TLR of the hematopoietic stem cells and induced them to proliferate. They also discovered that while the stimulus promoted proliferation, it also induced stress on the stem cells at the same time. In other words, although hematopoietic stem cells proliferate temporarily upon TLR stimulation, their ability to successfully self-replicate decreases, resulting in diminished blood production. Similar results were obtained after infection with E. coli (salmonella) as well.
Future Work
This study reveals that hematopoietic stem cells, while not in charge of immune reactions, are able to respond to bacterial infections resulting in a reduced ability to produce blood. This suggests that cell division of hematopoietic stem cells forced by bacterial infection induces stress and may further cause dysregulated hematopoiesis like that which occurs in anemia or leukemia.
"Fortunately we were able to confirm that this molecular reaction can be inhibited by drugs," said one of the study leaders, Professor Hitoshi Takizawa of Kumamoto University’s IRCMS. "The medication maintains the production of blood and immune cells without weakening the immune reaction against pathogenic bacteria. It might be possible to simultaneously prevent blood diseases and many bacterial infections in the future."
This finding was posted online in "Cell Stem Cell" on 21 July 2017, and an illustration from the research content was chosen as the cover of the issue.
[Resource}
Takizawa, H.; Fritsch, K.; Kovtonyuk, L. V.; Saito, Y.; Yakkala, C.; Jacobs, K.; Ahuja, A. K.; Lopes, M.; Hausmann, A.; Hardt, W.-D.; Gomariz, Á.; Nombela-Arrieta, Cé. & Manz, M. G., Pathogen-Induced TLR4-TRIF Innate Immune Signaling in Hematopoietic Stem Cells, Promotes Proliferation but Reduces Competitive Fitness, Cell Stem Cell, Elsevier BV, 2017. DOI: 10.1016/j.stem.2017.06.013
[Paper Info]
TITLE:
Pathogen-induced TLR4-TRIF innate immune signaling in hematopoietic stem cells promotes proliferation but reduces competitive fitness
AUTHORS:
Hitoshi Takizawa, Kristin Fritsch, Larisa V. Kovtonyuk, Yasuyuki Saito, Chakradhar Yakkala, Kurt Jacobs, Akshay K. Ahuja, Massimo Lopes, Annika Hausmann, Wolf-Dietrich Hardt, Alvaro Gomariz, Cesar Nombela-Arrieta and Markus G. Manz
JOURNAL:
Cell Stem Cell
DOI:
http://dx.doi.org/10.1016/j.stem.2017.06.013
URL:
http://www.sciencedirect.com/science/article/pii/S1934590917302394
URL to Cell Stem Cell (pay wall)
http://www.cell.com/cell-stem-cell/fulltext/S1934-5909(17)30239-4
[Fund]
To H.T.:
KAKENHI from the Japanese Society of the Promotion of Science, (15H01519) the Kanae Foundation for the Promotion of Medical Science, the SENSHIN Medical Research Foundation,
to C.N.-A.
the Swiss National Science Foundation, (31003A_159597/1)
to M.G.M.
the Swiss National Science Foundation, (310030B_166673/1) the Promedica Foundation (Chur, Switzerland), the Clinical Research Priority Program Human Hemato-Lymphatic Diseases of the University of Zurich
[image1]

Bacterial infection decreases ability to produce blood
CAPTION: Bacterial infection activates hematopoietic stem cells in the bone marrow and significantly reduces the ability to produce blood through induced proliferation.
[image2]
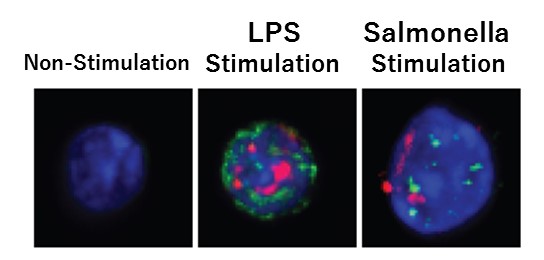
Stress on stem cells increases during bacterial infection
CAPTION: Blue shows DNA, red and green shows stress on the hematopoietic stem cell.[CREDIT] Professor Hitoshi Takizawa
[USAGE RESTRICTIONS] This image may only be used in conjunction with the accompanying release, or stories written about the work described in the release with reference to the original work.
