Novel lncRNA, Caren, counteracts heart failure progression

A research collaboration based in Kumamoto University has identified a novel lncRNA, Caren, that is abundantly expressed in cardiomyocytes. They showed that it enhances energy production by increasing the number of mitochondria in cardiomyocytes, and inhibits activation of the ATM protein, a key player in the DNA damage response pathway that accelerates heart failure severity. Caren RNA in cardiomyocytes is reduced by aging and high blood pressure (hypertension), which can lead to heart failure, and markedly reduced in the hearts of heart failure patients. The researchers believe that activation of Caren in cardiomyocytes could lead to the development of new heart failure therapies.| Heart failure mechanisms due to decreased Caren RNA in cardiomyocytes |
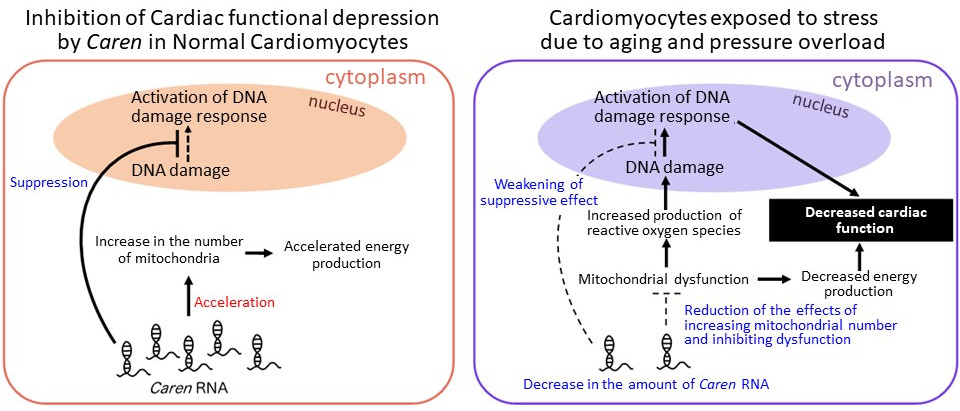 |
| Caren RNA is abundant in normal cardiomyocytes. It prevents deterioration of cardiac function by enhancing energy production through increased mitochondrial number and inhibiting the activation of the DNA damage response pathways. However, Caren RNA is reduced in cardiomyocytes that are exposed to aging and pressure stress. This leads to mitochondrial dysfunction and activation of the DNA damage response resulting in the development and exacerbation of heart failure. |
Heart failure is when reduced pumping function (contraction and dilatation) of the heart muscle is unable to pump enough blood to the body. It is still a disease with a poor prognosis and the number of heart failure patients is increasing worldwide. In developed countries, the increase in the number of heart failure patients, especially in the elderly, is a major problem. Therefore, there is a need to develop effective new treatment strategies.
Energy production from mitochondria is essential for maintaining cardiac function. Aging and hypertension, which increase the chances of heart failure development, cause mitochondrial dysfunction in cardiomyocytes that results in reduced mitochondrial energy production and increased reactive oxygen species production. Reactive oxygen species cause DNA damage, which subsequently activates the DNA damage response resulting in the exacerbation of heart failure. Therefore, mitochondrial dysfunction and the activation of the DNA damage response have both attracted attention as a cause of heart failure.
A research group led by Professor Oike at Kumamoto University has identified a novel lncRNA abundantly expressed in mouse cardiomyocytes and named it Caren (cardiomyocyte-enriched noncoding transcript). They also found that the amount of Caren RNA in mouse cardiomyocytes is reduced by stress, which can lead to heart failure. Further analysis of the function of Caren in the mouse heart revealed that it inhibits the decline of cardiac pump function. The researchers thus suggested that aging and stress reduce the amount of Caren RNA in cardiomyocytes, thereby reducing its effects and promoting mitochondrial dysfunction and activation of the DNA damage response, leading to the development and worsening of heart failure.
| Effect of Caren RNA supplement therapy on myocardial cells in the progression of heart failure |
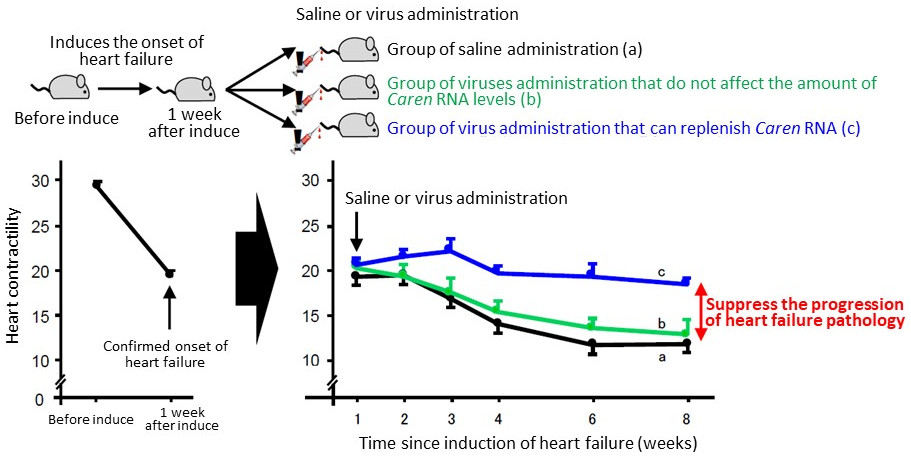 |
| In mice with artificially induced heart failure, the contractility of the heart was markedly reduced after one week which indicated the onset of heart failure. Researchers then administered saline (a; black), a virus that does not affect the amount of Caren RNA (b; green), or a virus that can replenish Caren RNA (c; blue) to the mice that had developed heart failure, and analyzed the changes in cardiac contractility. In the groups that received saline (a; black) and the virus that did not affect the amount of Caren RNA (b; green), the contractility of the heart continued to decrease and heart failure continued to progress. However, in the group that received the Caren RNA replenishing virus (c; blue), decreased heart contractility was suppressed. This suggests that Caren RNA replenishment therapy is effective in suppressing the progression of heart failure. [Adapted from Fig. 8d of Sato, M. et.al., Nature Communications, 2020. This image is made available under the terms of CC by 4.0. Any further distribution of this work must follow the terms of this license.] |
The researchers then genetically engineered a non-pathogenic adeno-associated virus to selectively infect cardiomyocytes and express Caren. After infecting heart failure model mice with the virus, they found that the amount of Caren RNA in cardiomyocytes increased, the number of mitochondria increased, and activation of the DNA damage response was suppressed compared to mice that were infected with a control virus, thus inhibiting the progression of heart failure in mice. The researchers also found that Caren RNA is present in human cardiomyocytes, and that its amount is inversely correlated with the expression level of heart failure marker genes in the heart tissue of heart failure patients. (The expression level of heart failure marker genes is high in heart tissues with low Caren RNA levels). Furthermore, they showed that decreasing the amount of human Caren RNA in cardiomyocytes generated from human iPS cells decreased the energy production capacity of mitochondria.
"Our research shows that increasing the amount of Caren RNA in cardiomyocytes can inhibit the onset and progression of heart failure, which we expect can be a strategy for developing new heart failure therapies," said Professor Oike. "In our in vivo mouse experiments, we found that Caren RNA supplement therapy using an adeno-associated virus was effective in counteracting heart failure progression. Now, we are going to verify whether human Caren has the same effect which could lead to the development of a new treatment for heart failure."
| Strategies for developing new heart failure therapies |
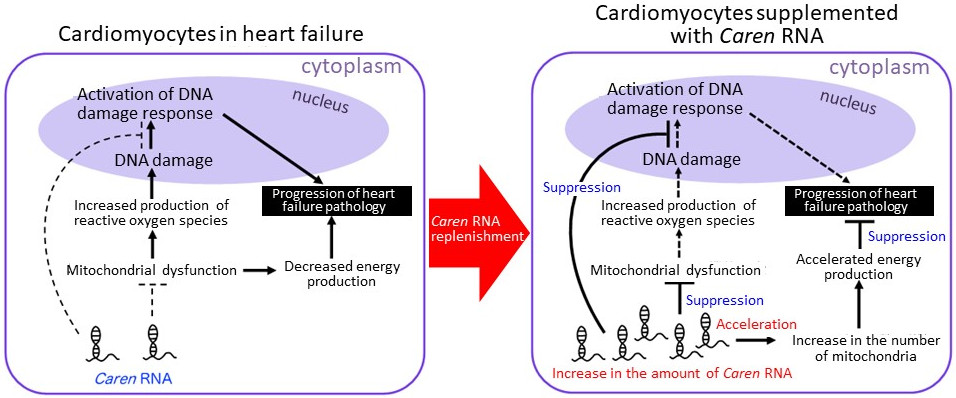 |
| Caren RNA levels are reduced in the cardiomyocytes of individuals suffering from heart failure. The resulting diminished effects lead to the progression of heart failure pathology. Supplementing cardiomyocytes with Caren RNA increases the number of mitochondria and promotes energy production through improved function. This can then inhibit both the activation of the DNA damage response and the exacerbation of heart failure pathology. Researchers expect this to be a new strategy in the development of heart failure therapies. |
This research was posted online in Nature Communications on 5 May 2021.
[Source]
Sato, M., Kadomatsu, T., Miyata, K., Warren, J. S., Tian, Z., Zhu, S., ... Oike, Y. (2021). The lncRNA Caren antagonizes heart failure by inactivating DNA damage response and activating mitochondrial biogenesis. Nature Communications, 12(1). doi:10.1038/s41467-021-22735-7
[Funding]
- Grants-in-Aid from MEXT Japan (JP18H02809, JP18K19519, JP19K17607, JP25461114, JP19K08560, JP23310135),
- Core Research for Evolutional Science and Technology (CREST) Program of the Japan Science and Technology Agency (JST) (13417915),
- CREST Program of the Japan Agency for Medical Research and Development (AMED) (18gm0610007),
- Project for Elucidating and Controlling Mechanisms of Aging and Longevity (AMED) (19gm5010002),
- SENSHIN Medical Research Foundation,
- Grant for Basic Research of the Japanese Circulation Society (2020),
- Harold S. Geneen Charitable Trust Awards Program for Coronary Heart Disease Research,
- Nora Eccles Treadwell Foundation,
- NIH Grant (R01HL130424)
[Usage Restrictions]
Figure credits belong to Professor Yuichi Oike unless otherwise indicated at the bottom of the figure caption. To use any of the media contained within this release elsewhere, a reference to the original work (this release) should be included and other restrictions as indicated must be followed.
