Atomic-level, 3-D structure of MUTYH protein opens small window into DNA repair mechanism

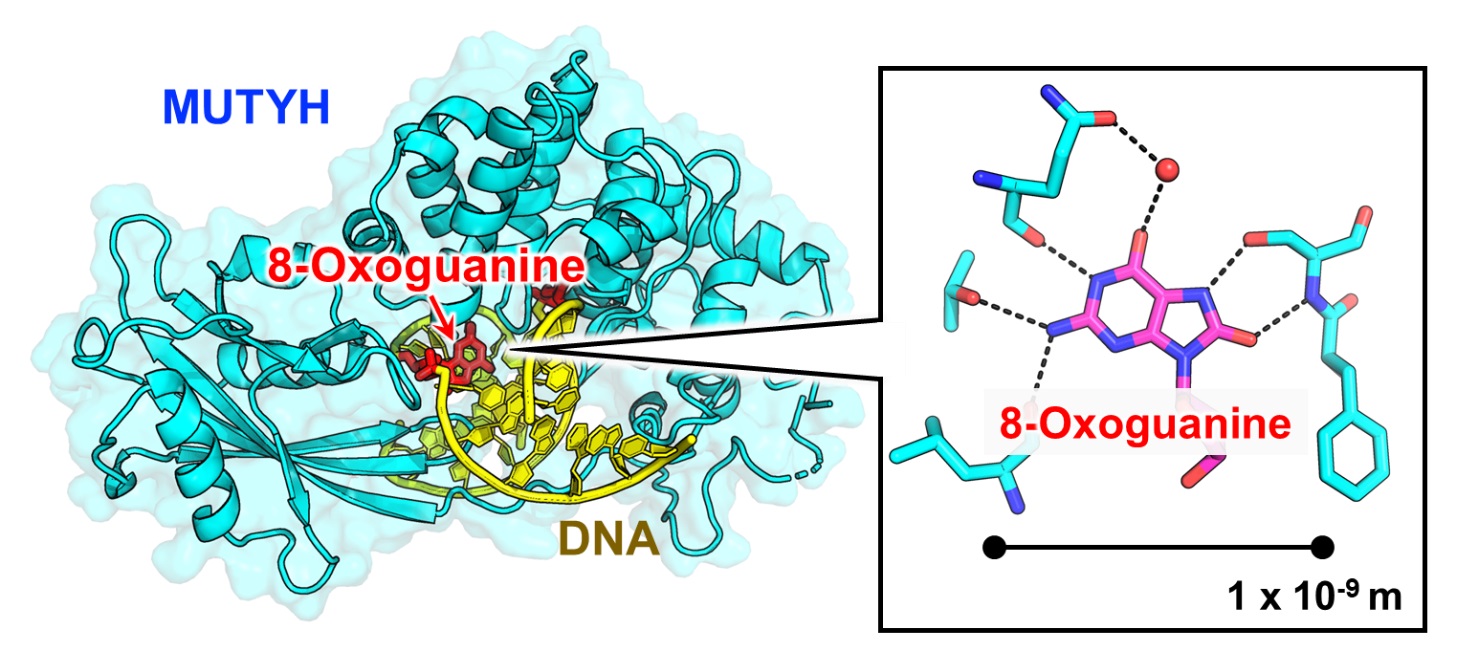
Researchers have analyzed the three-dimensional structure of a protein that suppresses the development of colorectal polyposis, MUTYH, at the atomic level and clarified the repair mechanism for DNA mispairings by MUTYH. Since mutations in the MUTYH gene cause heritable colorectal polyposis, which leads to colorectal cancer, the researchers expect that this work will be useful for future research on heritable colorectal polyposis associated with MUTYH.Reactive oxygen species produced inside cells oxidize DNA, and when the guanine base in DNA undergoes oxidation, 8-oxoguanine base is produced. Normally, guanine pairs with cytosine, but 8-oxoguanine also pairs with adenine, causing mutations that can lead to cancer and other aging-related diseases.
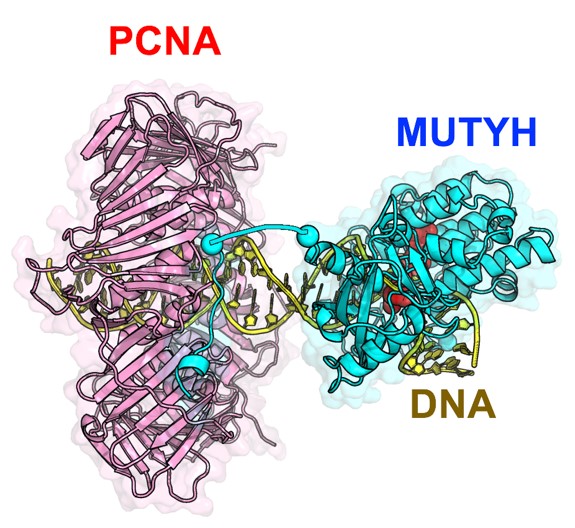
MUTYH is a protein that finds and removes adenine that has been mispaired with 8-oxoguanine. Mutations in its gene are known to cause heritable colorectal polyposis which can lead to colorectal cancer. Furthermore, the repair efficiency of MUTYH increases when it interacts with PCNA, a protein involved in DNA replication, but the repair mechanisms at the atomic level have not been clarified.
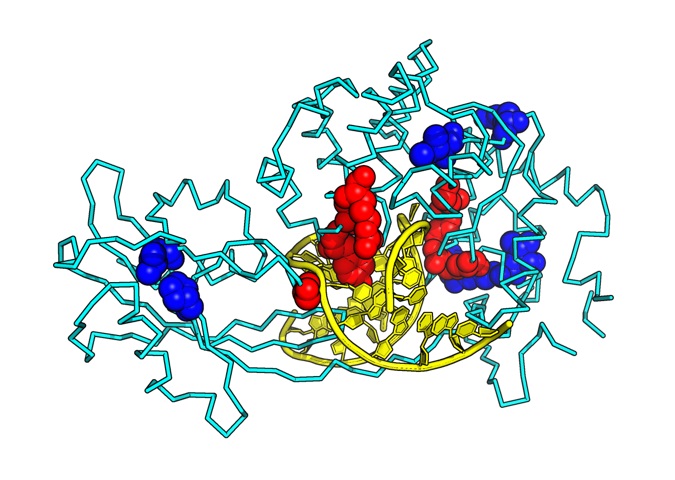
To accurately understand the function of a protein, it is important to know its structure at the atomic level. X-ray crystallography is a method that allows us to observe the molecules of living organisms at a scale of 1 x 10-10 m. Using this method, a research group led by Kumamoto University (Japan) has determined the X-ray crystal structure of MUTYH bound to a mispairing in DNA and the X-ray crystal structure of MUTYH bound to PCNA. Their analysis showed how MUTYH binds to the DNA double helix and searches for adenine:8-oxoguanine mispairings in DNA. Based on the three-dimensional structures of PCNA and MUTYH, the researchers proposed that PCNA serves as a clamp on the DNA double helix and recruits MUTYH to the mispair site, which would clarify the mechanism by which MUTYH and PCNA cooperate to repair DNA. Furthermore, analysis of the 3D structure of MUTYH and DNA showed that mutations in the MUTYH gene reduce the binding affinity of MUTYH to DNA and destabilize the 3D structure of MUTYH, which leads to a decrease of its DNA repair activity.
"Since MUTYH and PCNA are known to work with various proteins involved in DNA repair, the three-dimensional structures that we clarified in this study should serve as a basis for further understanding the DNA repair mechanisms centered around MUTYH and PCNA," said Associate Professor Teruya Nakamura, who led the study. "We expect that the clarification of their atomic-level structures will be useful for future studies of heritable colorectal polyposis associated with MUTYH."
This research was posted online in Nucleic Acids Research on 18 June 2021.
[Source]
Nakamura, T., Okabe, K., Hirayama, S., Chirifu, M., Ikemizu, S., Morioka, H., Nakabeppu, Y., Yamagata, Y. (2021). Structure of the mammalian adenine DNA glycosylase MUTYH: insights into the base excision repair pathway and cancer. Nucleic Acids Research. doi:10.1093/nar/gkab492
[Funding]
- Leading Initiative for Excellent Young Researchers from MEXT Japan,
- Grants-in-Aid for Scientific Research from MEXT Japan (25460038),
- Grant-in-Aid for Scientific Research on Innovative Areas from MEXT Japan (22131007),
- Grant for Basic Science Research Projects from The Sumitomo Foundation,
- Takeda Science Foundation,
- Kato Memorial Bioscience Foundation,
- KUMAYAKU Alumni Research Foundation
[Usage Restrictions]
Image credits belong to Dr. Teruya Nakamura unless otherwise indicated in the figure caption. To use any of the media contained within this release elsewhere, a reference to the original work (this release) should be included and other restrictions must be followed as indicated.