Japanese Scientists May Have Unraveled the Secret of Aging-Resistance in Naked Mole-Rats

Researchers from Japan identify a unique species-specific mechanism used by naked mole-rats to resist age-related deterioration and diseases
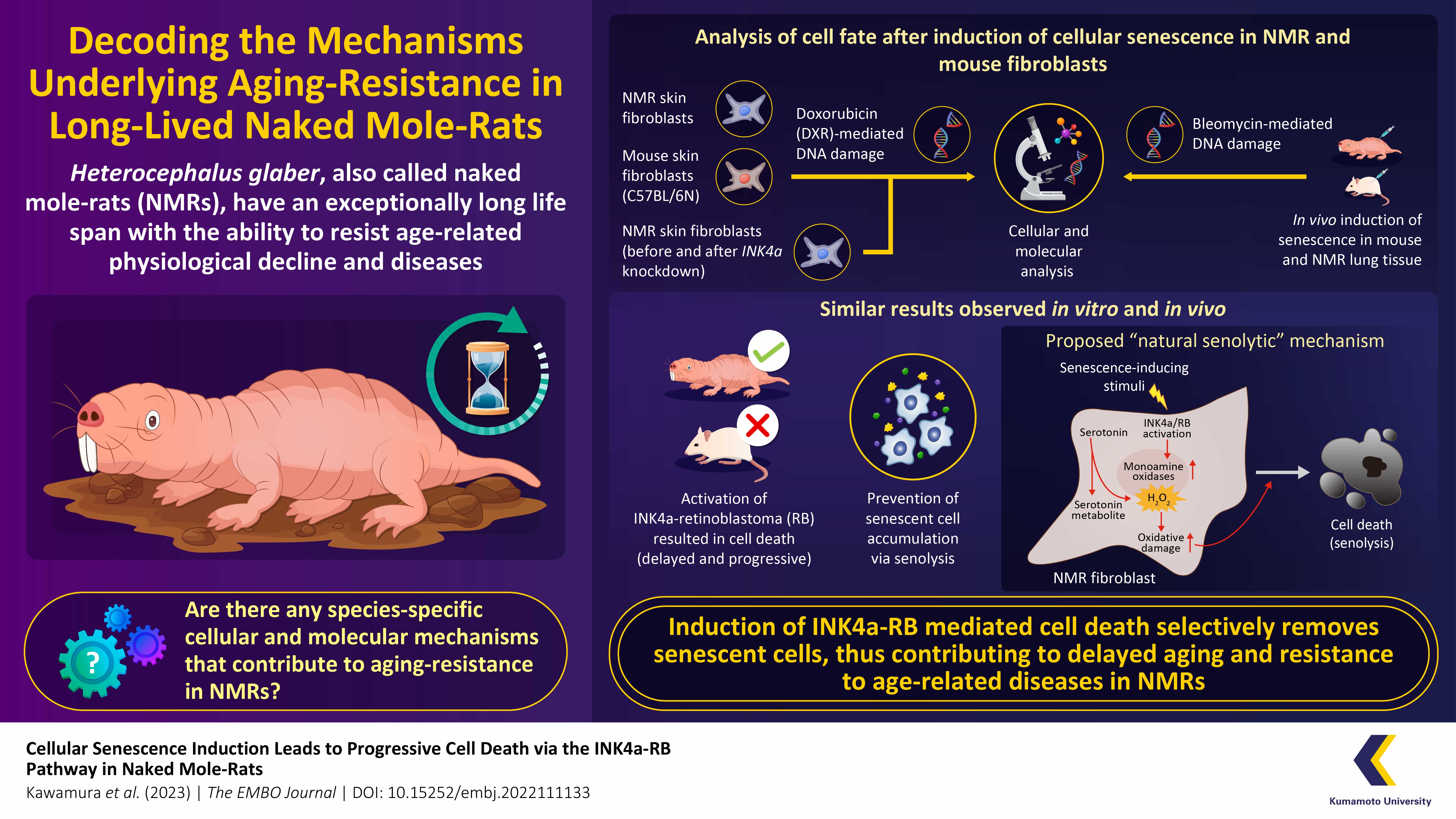
Naked mole-rats (NMRs) have the longest life span among all rodents, over 30 years, and can resist aging and the age-related diseases. However, the precise mechanisms underlying this ability are largely unclear. In a new study, Japanese researchers have identified a unique species-specific “natural senolytic” or senescent cell-removal mechanism in NMRs, involving serotonin metabolism and the INK4a-RB signaling axis. Their findings provide useful insights into ways to resist aging and age-related diseases, including cancer.
Heterocephalus glaber or naked mole-rats (NMRs)—a mammal species native to Eastern Africa—are the longest-living rodents with an exceptionally long lifespan of over 37 years with a unique ability to delay aging and resist age-related diseases, such as cancer. For these reasons, NMRs have attracted a lot of attention, with researchers hoping to unravel the mechanisms contributing to their longevity. Previous studies have explored the role of DNA repair mechanisms, protein stability, and translation accuracy (precise conversion of RNA to proteins) in this regard, but the molecular mechanisms/factors behind their aging-resistance remain largely unclear. Moreover, the contribution of cellular senescence to their aging resistance is poorly understood.
Cellular senescence (cellular aging) is characterized by the irreversible arrest of cell division, which progresses with age. Senescent cells are less prone to cell death and accumulate in the tissues as they age, promoting chronic inflammation and compromising the function of these tissues. While cellular senescence plays an important role in aging, little is known about its function in NMRs.
To this end, a team of researchers from Japan led by Professor Kyoko Miura from the Department of Aging and Longevity Research, Kumamoto University, conducted a series of experiments in vitro and in vivo to understand how cellular senescence occurs in NMRs and if there are any species-specific mechanisms that contribute to suppress accumulation of senescent cells and their delayed aging. The Department of Aging and Longevity Research, Kumamoto University is the only center in Japan which breeds NMRs and conducts research on their resistance to aging and cancer. Explaining the rationale behind their study published on July 11, 2023 in The EMBO Journal, Professor Miura, states, “Senolysis or the targeted removal of senescent cells has been shown to inhibit aging-related decline in mice. However, whether the findings in mice are generalizable, remains an open question. In this study, we discovered an NMR-specific “natural senolytic” mechanism that may provide an evolutionary rationale for removing senescent cells as a therapeutic strategy to prevent aging.”
The research team used low concentrations of doxorubicin (DXR)—a DNA damaging agent— to induce cellular senescence in NMR- and mouse-derived skin fibroblasts in vitro. They observed that induction of cellular senescence led to cessation of cell proliferation due to arrest of the cell cycle with the activation of INK4a and RB (important factors for induction of cellular senescence), in both NMR- and mouse-fibroblasts. However, only NMR cells gradually and significantly activated cell death, suggesting that senescent cell accumulation in NMRs may be suppressed through their removal.
Through further experiments, the researchers observed that there was an accumulation of serotonin (a neurotransmitter that sends signals between nerve cells) in the non-senescent NMR-fibroblasts, but not in the mouse-fibroblasts. Upon senescence induction, in NMR cells, serotonin was metabolized by monoamine oxidase (MAO; an enzyme highly activated in senescent NMR fibroblasts after induction of cellular senescence) and converted to 5-hydroxyindole acetic acid (5-HIAA; a metabolite), releasing large amounts of hydrogen peroxide (H2O2). The team proposed that oxidative stress due to the intracellular production of H2O2 predisposed the senescent NMR fibroblasts to the cell death pathway, thus leading to senolysis (selective removal of senescent cells). This was confirmed by the observation that the addition of MAO inhibitors and antioxidants inhibited cell death in NMR fibroblasts.
To confirm if a similar mechanism was also prevalent in vivo, the team induced cellular senescence in the lungs of mice and NMRs using bleomycin (a DNA damaging agent). They observed that cell death, likely due to an acute response to DNA damage, initially increased on day 2 in both mouse and NMR lung cells. However, after an initial rise on day 2, a fall in cell death was observed and by day 21, cell death had increased again, only in NMR lung cells. Furthermore, treatment with the MAO inhibitor significantly suppressed cell death but increased the number of senescent cells only in NMR lung on day 21. This suggests that MAO plays a role in inducing cell death and reducing the number of senescent cells following the induction of cellular senescence in NMR lung cells. These results are consistent with the in vitro findings and suggest that MAO contributes to suppress the accumulation of senescent cells in NMR tissues. “Further studies focusing on the senescent cell removal mechanism in NMR tissues are needed to understand which kind of senescent cells should be removed, when, and how. Such studies may aid the development of safer and targeted senolytic drugs,” says Prof. Miura while discussing future steps.
Overall, these findings suggest that INK4a-RB-mediated cell death may facilitate the removal of senescent cells in NMRs, helping them resist aging-related degeneration. We are confident that by highlighting a natural senolytic mechanism in this long-lived species, this study would contribute to the development of anti-aging strategies and targeted therapies against age-related diseases such as cancer.
About Professor Kyoko Miura from Kumamoto University
Professor Kyoko Miura is a faculty member at the Department of Aging and Longevity Research, Kumamoto University, Kumamoto, Japan. Her research primarily focuses on elucidating the mechanisms underlying resistance to cancer and aging in NMRs. Along with her team, Prof. Miura aims to identify the molecular mechanisms and genes that contribute to the resistance to aging and cancer in NMRs, and to understand their relationship with evolutionary adaptation to their natural habitat. Prof. Miura also aims to extrapolate findings from the NMR model to several disease models of laboratory mice and, potentially, human diseases in the future. She is a member of several international research societies and has been recognized for her exemplary work, which has been published in various peer-reviewed journals.
Media contact: miurak@kumamoto-u.ac.jp